LightChEC: Solar Light to Chemical Energy Conversion
(contact Prof. Juerg Osterwalder,
osterwal@physik.uzh.ch)
The University Research Priority Program LightChEC is a joint project between the Department of Chemistry and
Department of Physics of the University of Zurich and the
EMPA in Dübendorf.
The objectives of the project are the discovery and development of new molecules, materials and processes
for the direct storage of solar light energy in chemical bonds, prominently via the splitting of water into hydrogen and oxygen gas.
The heterogeneous route to a solar-light-driven water splitting process is technologically compelling because the water reduction and oxidation steps can be spatially separated, thus avoiding unwanted recombination reactions and facilitating separation of the produced gaseous hydrogen and oxygen. In a photoelectrochemical (PEC) cell, each of the two electrodes can be optimized for the respective reaction. Typically, they consist of a semiconducting material with a small enough band gap to absorb a large fraction of the solar spectrum. Upon irradiation electron-hole pairs are created that need to be separated efficiently in order to avoid recombination. At semiconductor interfaces, band bending can introduce electrical fields that steer the proper charge to the electrode-electrolyte interface. Finally, these charges need to be energetic enough and live long enough at the interface to drive the respective redox-reaction, which is typically promoted by a catalyst. Last but not least, the electrode surface has to be stable under these rather harsh conditions.
Characterizing and understanding individual steps of this complex sequence of events may help to optimize the overall efficiency of PEC cells. In our group we prepare model electrodes based on materials and catalysts that are synthesized and used by other groups within the LightChEC consortium. Our emphasis is on single-crystalline material where we can have precise control over composition, structural parameters and electronic energy levels using a versatile toolbox of surface science techniques. In two subprojects we study such model systems either under ultrahigh vacuum (UHV) environment (Surface Science and Ultrafast Electron Dynamics) or under near-ambient pressure conditions (In-Situ Spectroscopy):
Subproject 1: Surface Science and Ultrafast Electron Dynamics (discontinued)
(contact Dr. Matthias Hengsberger,
matthias.hengsberger@physik.uzh.ch)
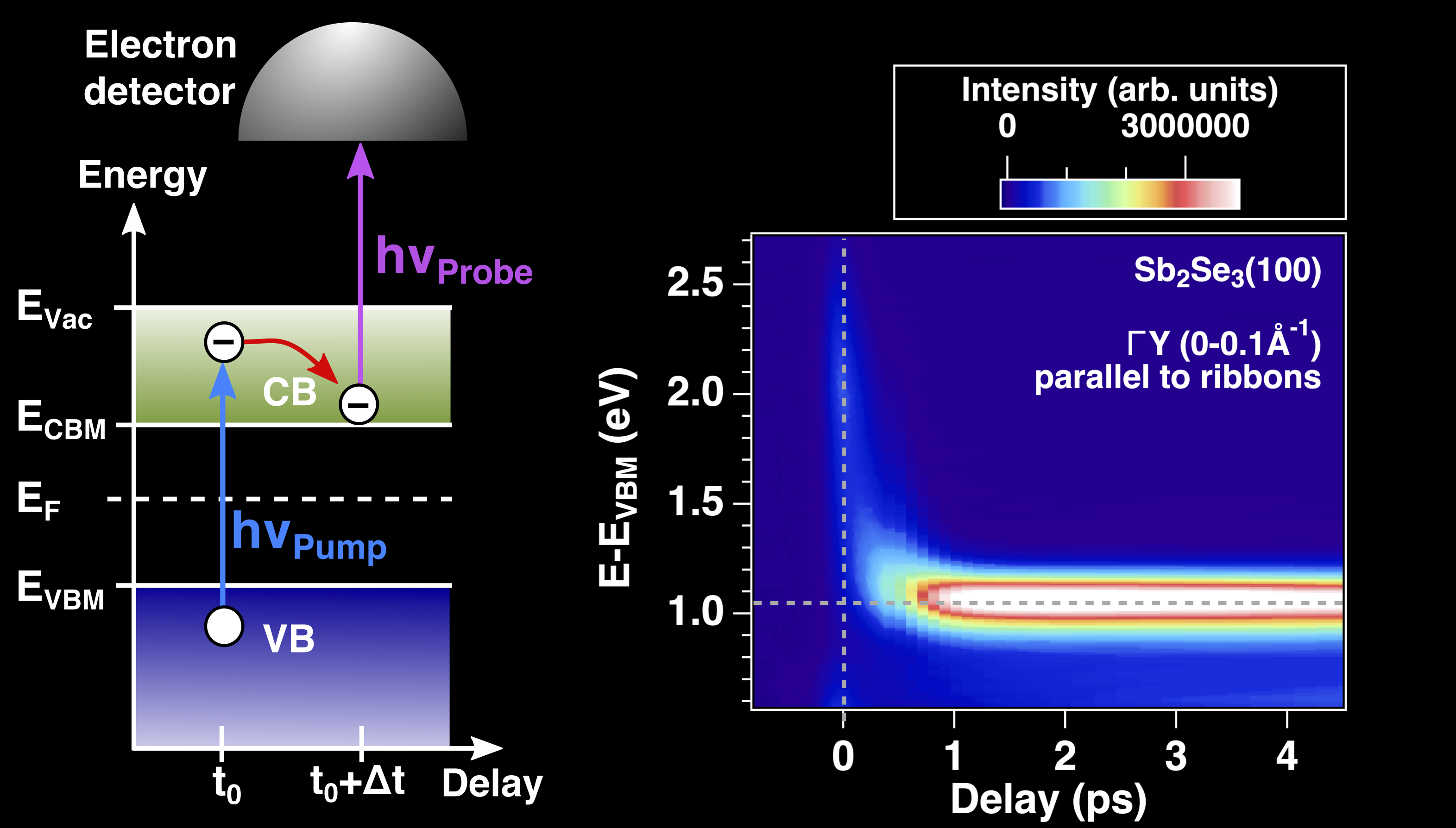
Moreover, time-resolved two-photon photoemission provides information on the dynamics of hot electrons after excitation by ultrashort laser pulses (see Fig. 1).
Two photoelectron spectrometer chambers connected to a femtosecond laser system could be used for time-resolved two-photon photoemission (2PPE) installed in the adjacent laboratory.
Subproject 2: In-Situ Spectroscopy
(This project will likely be continued by the groups of M. Ammann and J. van Bokhoven at PSI/SLS.)
In June 2018, a new endstation was installed at the Swiss Light Source. Designed and assembled by Ferrovac GmbH, we introduced a unique endstation that combines ultra-high vacuum sample preparation with ambient pressure X-ray photoelectron spectroscopy and X-ray absorption spectroscopy (XAS) analysis.
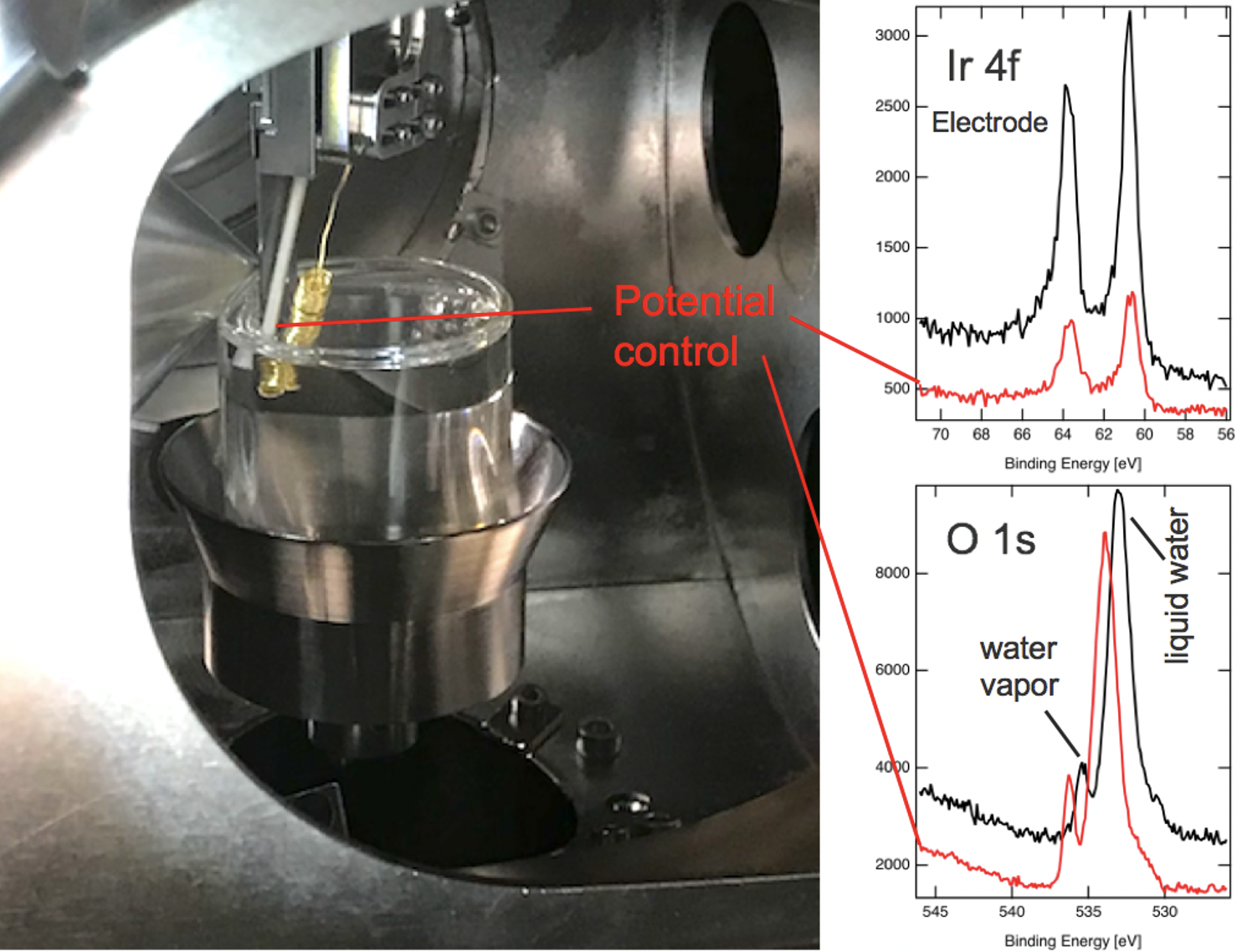
This ambient pressure X-ray photoelectron spectroscopy (APXPS) endstation (see Fig. 2) is a modular system where the electron analyzer (ScientaOmicron R4000 HiPP-2) can be connected to two experimental chambers. In-situ photoelectron spectroscopy experiments in the millibar range allow the characterization of different interfaces under relevant conditions

The endstation can be operated at the In-situ Spectroscopy beamline for soft X-ray experiments (pressure up to 5 mbar) and at the PHOENIX I tender X-ray beamline (pressure up to 40 mbar). A unique feature of this setup is its capability to probe liquid-solid interfaces under potential control and/or illumination with a solar simulator. Utilizing the dip and pull method, thin layers of electrolyte (10-30 nm) with potential control can be created and stabilized on top of a solid substrate. The buried solid-liquid interface is probed using tender x-rays, producing photoelectrons that can escape the thin liquid layer (see Fig. 3). Such experiments are performed at the Phoenix I beamline at the SLS. Up to now, we focused our research on well-defined metal oxides with rutile structure, such as TiO2(110), RuO2(110), IrO2(110).
The endstation was commissioned in the 2018-2019 period. The endstation is fully operational, with restricted access for general users. Our current research's primary focus is centered on in situ studies of the systems developed within the framework of the LightChEC program.
Team
- Dr. Matthias Hengsberger
- Theodor Schaller
Latest publications
- h-BN/Metal-Oxide Interface Grown by Intercalation: A Model System for Nano-Confined Catalysis
The Journal of Physical Chemistry C 128, 5156 (2024)
→ DOI: 10.1021/acs.jpcc.3c07828 - Revealing the Bonding Nature and Electronic Structure of Early-Transition-Metal Dihydrides
PRX Energy 3, 013003 (2024)
→ DOI: 10.1103/PRXEnergy.3.013003 - Surface electronic structure of Ni-doped Fe3O4(001)
Phys. Rev. B 108, 155403 (2023)
→ DOI: 10.1103/PhysRevB.108.155403 - Stability of Iridium Single Atoms on Fe3O4(001) in the mbar Pressure Range
J. Phys. Chem. C , 127, 19097 (2023)
→ DOI: 10.1021/acs.jpcc.3c03097 - Formation of NixFe(3-x)O4 on Fe3O4(001)
Phys. Rev. Materials 7, 055801 (2023)
→ DOI: 10.1103/PhysRevMaterials.7.055801 - ...[→ more publications]



