Physics on the Nanometer Scale
with a particular emphasis on the physics of biological systems
Group of Hans-Werner Fink

| ...last update: January 2016 |
| Science |
| Teaching |
| People - Contact |
| Movies |
| STARTPAGE |
OPEN POSITIONS:
none, right now
 diffraction pattern behind a helium-ion milled hole array structure in a free-standing thin carbon film, taken with 65eV coherent electrons
diffraction pattern behind a helium-ion milled hole array structure in a free-standing thin carbon film, taken with 65eV coherent electrons
Properties and Applications of Electron and Ion Point Sources |
We employ Field Ion Microscopy and related techniques for fabricating and using electron and ion point sources. Coherent low-energy electron beams are used to acquire electron holograms of individual biological molecules which can be numerically reconstructed. More recently, we combined dedicated electron optical devices with our coherent electron beams to obtain over sampled coherent diffraction patterns from a single molecule. We expect that both methods eventually lead to new tools for structural biology on a single molecule level, free from disturbing radiation damage effects inherent to other radiation with sufficient short wavelengths and thus potential for atomic resolution. For presenting a fragile single molecule to the coherent electron beam we structure free standing thin films with a focused gallium ion beam. Interfacing single molecules from the liquid phase to such nanometer-sized structures is another pre requisite before imaging by low energy electrons becomes possible. Fluorescent video microscopy allows us to observe and manipulate single DNA molecules already in the liquid phase and to explore the energetics of this important bio-polymer. Molecular biology expertise and tools for anchoring DNA and/or modifying the molecules according to our specific needs for imaging are provided to us by Clondiag Chip Technologies.
|
Some of the tools and methods we use are |
|
Field Ion and Field Electron Microscopy to produce and characterize electron and ion sources. Low Energy Electron Point Source (LEEPS) Microscopy for electron holography. Focused Liquid Metal Ion Beams for structuring thin films and creating micron-sized electron optical devices for Coherent Diffraction Imaging (CDI). Scanning Electron Microscopy (SEM) for sample characterisation. Fluorescent Video Microscopy for single DNA experiments in the liquid phase. read more
|
|
Major Achievements |
|
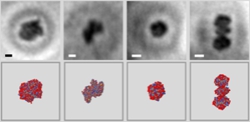
Imaging proteins at the single molecule level, by Jean-Nicolas Longchamp, Stephan Rauschenbach, Sabine Abb, Conrad Escher, Tatiana Latychevskaia, Klaus Kern, Hans-Werner Fink - PNAS, Early Edition: Friday, January 13th, 2017
All established methods for obtaining structural information about proteins require averaging over thousands or even millions of molecules, be it due to radiation damage or a too weak signal from just one individual protein. Since proteins are flexible objects that can assume distinct conformations often associated with different functions, averaging over a large ensemble of proteins leaves these associated structural details undiscovered.
Atomically resolved structural determination of graphene and its point defects via extrapolation assisted phase retrieval, by Tatiana Latychevskaia and Hans-Werner Fink, Appl. Phys. Lett. 106, 021908 (2015), read more Previously reported crystalline structures obtained by an iterative phase retrieval reconstruction of their diffraction patterns seem to be free from displaying any irregularities or defects in the lattice, which appears to be unrealistic. We demonstrate here that the structure of a nanocrystal including its atomic defects can unambiguously be recovered from its diffraction pattern alone by applying a direct phase retrieval procedure not relying on prior information of the object shape. Individual point defects in the atomic lattice are clearly apparent. Conventional phase retrieval routines assume isotropic scattering. We show that when dealing with electrons, the quantitatively correct transmission function of the sample cannot be retrieved due to anisotropic, strong forward scattering specific to electrons. We summarize the conditions for this phase retrieval method and show that the diffraction pattern can be extrapolated beyond the original record to even reveal formerly not visible Bragg peaks. Such extrapolated wave field pattern leads to enhanced spatial resolution in the reconstruction. Holography and coherent diffraction with low-energy electrons: A route towards structural biology at the single molecule level, by by Tatiana Latychevkaia, Jean-Nicolas Longchamp, Conrad Escher, Hans-Werner Fink, Ultramicroscopy (2014, online version), read more The current state of the art in structural biology is led by NMR, X-ray crystallography and TEM investigations. These powerful tools however all rely on averaging over a large ensemble of molecules. Here, we present an alternative concept aiming at structural analysis at the single molecule level. We show that by combining electron holography and coherent diffraction imaging estimations concerning the phase of the scattered wave become needless as the phase information is extracted from the data directly and unambiguously. Performed with low-energy electrons the resolution of this lens-less microscope is just limited by the De Broglie wavelength of the electron wave and the numerical aperture, given by detector geometry. In imaging freestanding graphene, a resolution of 2 Å has been achieved revealing the 660.000 unit cells of the graphene sheet from a single data set. Once applied to individual biomolecules the method shall ultimately allow for non-destructive imaging and imports the potential to distinguish between different conformations of proteins with atomic resolution.
Graphene Unit Cell Imaging by Holographic Coherent Diffraction, by Jean-Nicolas Longchamp, Tatiana Latychevskaia, Conrad Escher, Hans-Werner Fink, Phys. Rev. Lett. 110, 255501 (2013), read more We have imaged a freestanding graphene sheet of 210 nm in diameter with 2 Angstrom resolution by combining coherent diffraction and holography with low-energy electrons. The entire sheet is reconstructed from just a single diffraction pattern displaying the arrangement of 660.000 individual graphene unit cells at once. Given the fact that electrons with kinetic energies of the order of 100 eV do not damage biological molecules, it will now be a matter to develop methods for depositing individual proteins onto such graphene sheets. Ultra-Clean Freestanding Graphene by Platinum-Metal Catalysis by Jean-Nicolas Longchamp, Conrad Escher, Hans-Werner Fink, read more While freestanding clean graphene is essential for various applications, existing technologies for removing the polymer layer after transfer of graphene to the desired substrate still leave significant contaminations behind. We discovered a method for preparing ultra-clean freestanding graphene utilizing the catalytic properties of platinum metals. Complete catalytic removal of polymer residues requires annealing in air at a temperature between 175 and 350C. Low-energy electron holography investigations prove that this method results in ultra-clean freestanding graphene. Low-energy electron transmission imaging of clusters on free-standing graphene by Jean-Nicolas Longchamp, Tatiana Latychevskaia, Conrad Escher, Hans-Werner Fink, read more We investigated the utility of free-standing graphene as a transparent sample carrier for imaging nanometer-sized objects by means of low-energy electron holography. The sample preparation for obtaining contamination-free graphene as well as the experimental setup and findings are discussed. For incoming electrons with 66 eV kinetic energy graphene exhibits 27% opacity per layer. Hence, electron holograms of nanometer-sized objects adsorbed on free-standing graphene can be recorded and numerically reconstructed to reveal the object’s shapes and distribution. Furthermore, a Moiré effect has been observed with free-standing graphene multi-layers. See a movie, clip1, related to these findings Imaging of an individual protein by J.-N. Longchamp, T. Latychevskaia, C. Escher, and H.-W. Fink, read more Imaging a single biomolecule at atomic resolution without averaging over different conformations is the ultimate goal in structural biology. We report recordings of a protein at nanometer resolution obtained from one individual ferritin by means of low-energy electron holography. One single protein could be imaged for an extended period of time without any sign of radiation damage. Since the fragile protein shell encloses a robust iron cluster, the holographic reconstructions could also be cross-validated against transmission electron microscopy images of the very same molecule by imaging its iron core. Non-destructive imaging of individual biomolecules by Matthias Germann, Tatiana Latychevskaia, Conrad Escher & Hans-Werner Fink, read more Radiation damage is considered to be the major problem that still prevents imaging an individual biological molecule for structural analysis. So far, all known mapping techniques using sufficient short wavelength radiation, be it x rays or high energy electrons, circumvent this problem by averaging over many molecules. Averaging, however, leaves conformational details uncovered. Here, we present direct experimental evidence for nondestructive imaging of individual DNA molecules. In fact, we show that DNA withstands coherent low energy electron radiation with deBroglie wavelength in the Ångstrom regime despite a vast dose of 100 million electrons/square-nanometer accumulated over more than one hour. See a movie, clip2, related to these findings Electrons reveal DNA without destroying it, Physics World Magazine by James Dacey
|